https://link.springer.com/article/10.1007/s10689-023-00347-y
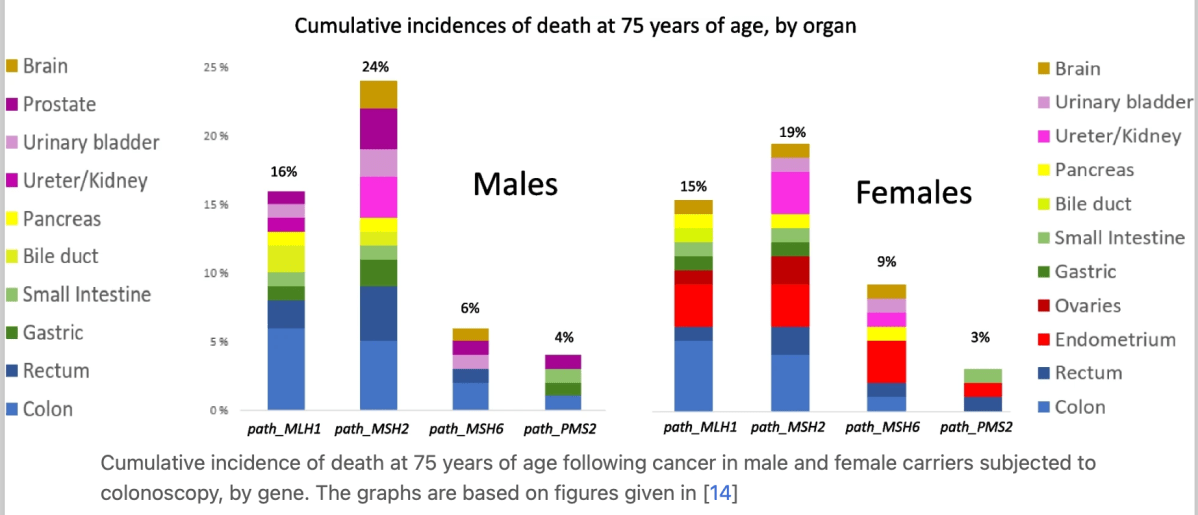
Evidenced-based modelling studies of gene and gender stratified populations suggest a start age and frequency later and less frequent, for MSH6 and PMS2 variant carriers (35 years and 2–3 yearly). The penetrance of MLH1 and MSH2 variant carriers on the other hand does not allow the later start time at least as agreed in most guidelines. This differential surveillance by gene is now being implemented in various guidelines around the world. Such prescription however needs prudent implementation given variable intragenic and even intravariant penetrance estimates in large studies. Note that the confidence intervals in these estimates is relatively large.
There is still the “here and now” which needs to engage carriers and optimize adherence to whatever scheduling is prudent and advised.
How do we do this?
First, registries. Registries keep track of carriers, can provide automated time alerts for the need to attend scheduling ….Registries save lives.
Secondly, patient navigators. Time and time again it has been shown that navigators personally chaperone carriers through the complexities of the healthcare system, to actually reach colonoscopy. They are worth their cost-benefit weight in gold at least in the syndromic scenarios we are discussing.
Thirdly, a pleasant experience helps maintain adherence – the least offensive and effective bowel preparation, a comfortable colonoscopic experience (sedation is important for adherence), and compassionate staff. Efficient throughput (less time for patients) on the day is also valuable. Bowel preparation regime should be personalized against previous experience.
Fourthly, the governance of the risk management service needs to be comprehensive (not just gastrointestinal), patient-centred, interactive with the patient navigators, general practitioners. associated familial cancer clinics (not losing opportunities for time and age appropriate cascade testing across the family), and hospital electronic medical records.
The current study amongst others is important if we are to benefit from the wealth of science that has been uncovered in the understanding of familial cancer. Minimizing the burden of colonoscopies is an important variable, warranting that debate herein described.