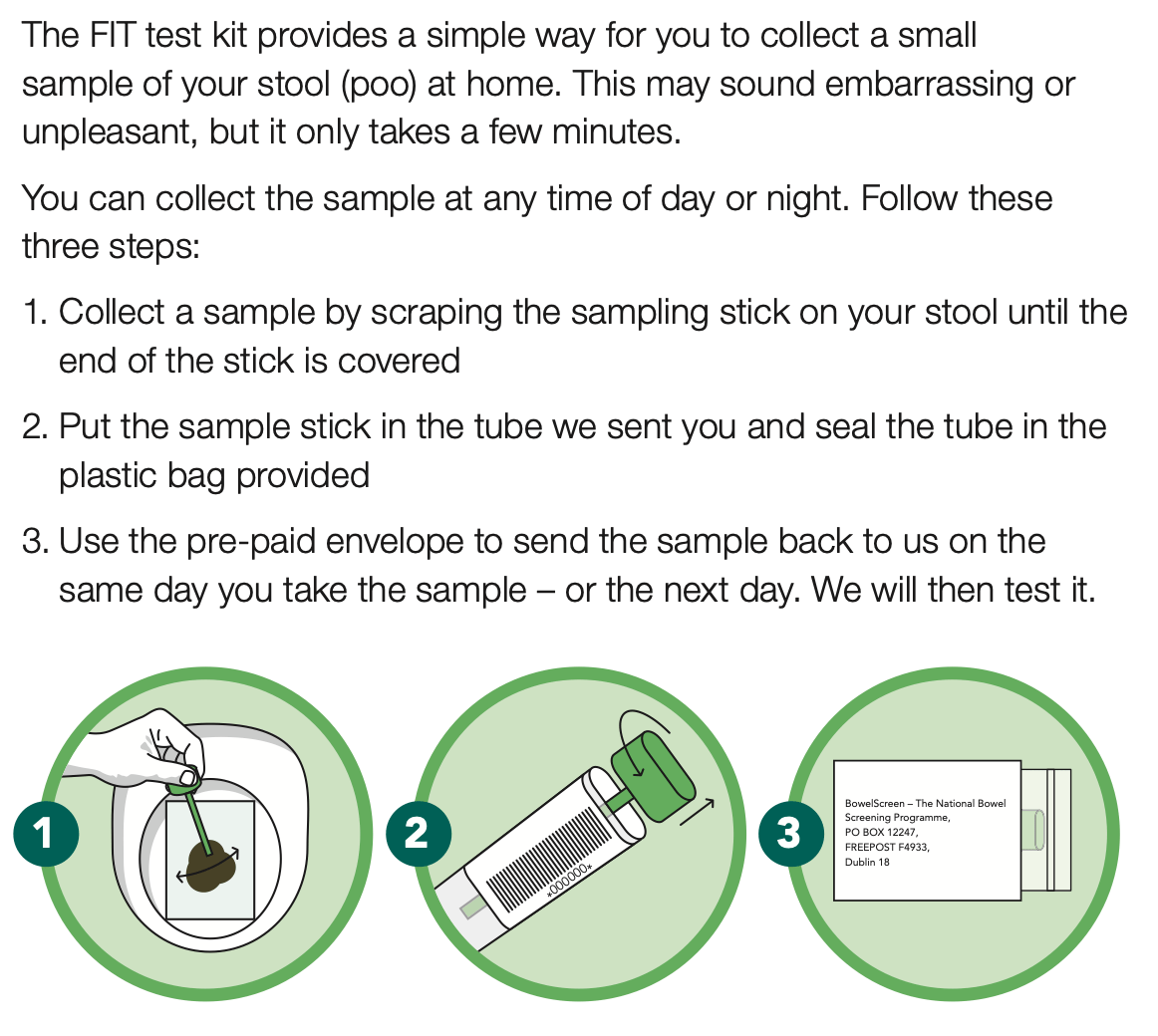
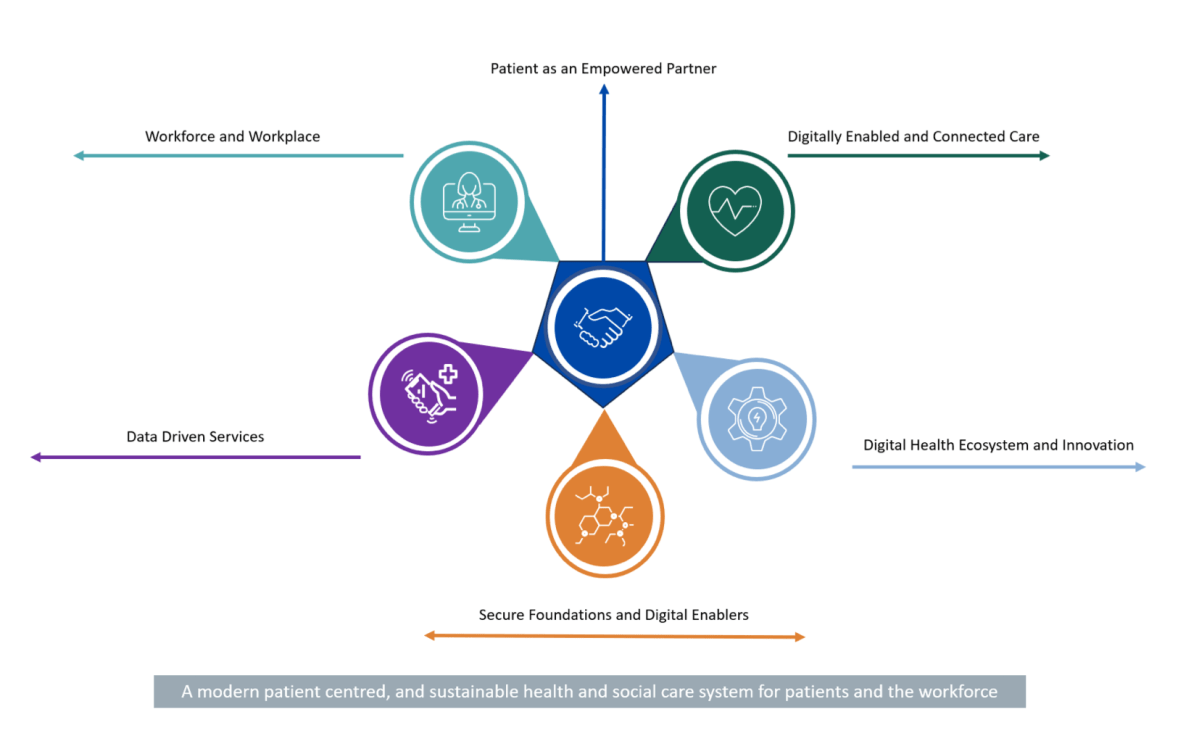
-Patient as an empowered partner: We will empower patients by giving them broader access to their own health information through a patient app, provide access to more digital health services, including virtual care offerings, whilst enabling greater autonomy and choice over their care options.
•Workforce and workplace: We will enable our workforce by providing them with the technology, systems, and skills they need to deliver the best possible care and services to patients in the modernised healthcare system.
•Digitally Enabled and Connected Care: We will drive future investment and make architectural decisions based on the ability of systems to share clinical information and deliver connected care. We will make it possible for healthcare professionals, and others who support delivery of care, to be able to access the information needed about their patients when and where they need it, regardless of where those patients were treated previously.
•Data driven services: We will leverage data analytics, business intelligence, visualisation, dashboards, and other digitally enabled management tools to provide greater insights into the health service and its operation. This will create opportunities for increased productivity, efficiency and more precise direction of resources to areas of greatest need, and where they will have the maximum impact.
•Digital health ecosystem & innovation: We will embed continuous improvement within the health and social care system. This will be enabled by innovation via improving ongoing collaboration, improved procurement pathways, increased participation, and promoting research excellence. This strategic principle also details key considerations for the Digital health ecosystem as we prepare for the technologies of the future.
•Secure foundations & digital enablers: We will continue to build cyber resilience and put in place the key enablers needed to deliver this digital health strategic roadmap, underpinned by strong governance, cultural change, standards, interoperability, infrastructure, architecture, and legislation.
https://www.gov.ie/en/publication/0d21e-digital-for-care-a-digital-health-framework-for-ireland-2024-2030/