Author: Lynch Syndrome Ireland
Tips for talking to someone with Cancer
In an Ideal World: We would have….
- A central point to support people who are affected by Lynch Syndrome or other Genetic Cancer defects.
- An integrated Cancer Genetics service.
- An increased Genetics workforce.
- A Genetics Testing center in Ireland.
- A comprehensive IT system to Manage/Track and generally improve the collection/storage of relevant information.
- Improved awareness by Public and Medics of Genetics.
- A dedicated Pathway for people with a genetic cancer issue.
- Improved support available to help in relaying a diagnosis to the wider family.
- A national BioBank to improve/help research in this area.
Blood Clots
Cancer patients, especially those who are receiving chemotherapy, have a much higher risk of DVT than other people.
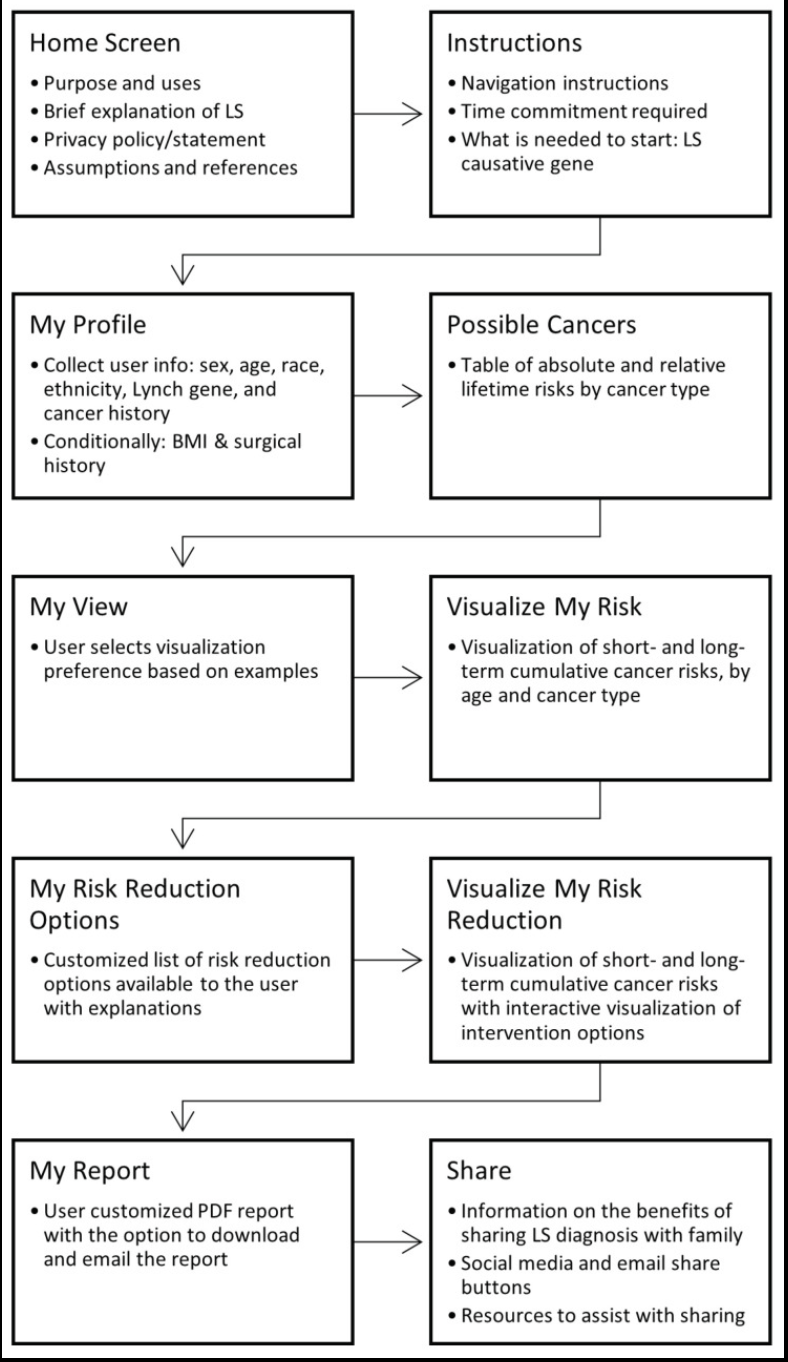
MyLynch: A Patient-Facing Clinical Decision Support Tool for Genetically-Guided Personalized Medicine in Lynch Syndrome (USA)
MyLynch, a patient-facing clinical decision support(CDS) web application that applies genetically-guided personalised medicine(GPM) for individuals with Lynch syndrome.
As genetic panel testing becomes more widely available, GPM will play an increasingly important role in patient care, and CDS tools offer patients and providers tailored information to inform decision-making.
MyLynch provides personalised cancer risk estimates and interventions to lower these risks for patients with LS.
A mainstreaming oncogenomics model: improving the identification of Lynch syndrome
“Mainstreaming” is a proposed strategy to integrate genomic testing into oncology. The aim of this paper is to develop a mainstreaming oncogenomics model by identifying health system interventions and implementation strategies for mainstreaming Lynch syndrome genomic testing.
https://pubmed.ncbi.nlm.nih.gov/37305562/
The English National Lynch Syndrome Transformation Project(UK)
Lynch syndrome affects approximately 1 in 400 individuals and predisposes to multiple cancers including colorectal, endometrial, gastric, small bowel and other tumours. Although a common condition, it is estimated that only 5% of patients with LS are known in the UK.
Lifelong care of people diagnosed with this condition depends on awareness of who this population is.
There is consistent evidence of the cost-effectiveness and clinical benefit of a structured diagnostic pathway in patients with LS following a diagnosis of cancer linked to cascade testing in families.
Barriers to diagnosis and a manifest deficiency in care for those with LS has been described in the literature as a ‘diffusion of responsibility’.
An approach to deliver effective diagnosis is to develop ‘mainstreaming’ models whereby patients are offered constitutional genetic testing by their cancer treating teams locally, rather than relying on referral of eligible patients to tertiary services such as clinical genetics.
This has many possible advantages including shorter timescale to diagnosis, effective communication provided through an existing relationship between patients and their clinical teams, and ensuring that eligible patents access testing. This model is associated with high levels of acceptability for patients and clinicians, however relies on the development or new skills by cancer teams.
https://www.bsg.org.uk/clinical-resource/(sss)-english-lynch-syndrome-project
Variant of Uncertain Significance (abbreviated VUS)
When a person undergoes a genetic test it can identify a variant(change), but it is unclear whether that variant is actually connected to a health condition for example Lynch Syndrome.
https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2825808
Conclusions and Relevance In this cohort study of individuals undergoing genetic testing, the empirically estimated accuracy of pathogenic, likely pathogenic, benign, and likely benign classifications exceeded the certainty thresholds set by current variant classification guidelines, suggesting the need to reevaluate definitions of these classifications.
The relative contribution of various strategies to resolve VUS, including emerging machine learning–based computational methods, RNA analysis, and cascade family testing, provides useful insights that can be applied toward further improving VC methods, reducing the rate of VUS, and generating more definitive results for patients.
Multigene Panel Testing Yields High Rates of Clinically Actionable Variants Among Patients With Colorectal Cancer
Purpose:
Whether germline multigene panel testing (MGPT) should be performed in all individuals with colorectal cancer (CRC) remains uncertain. Therefore, we aimed to determine the yield and potential clinical impact of MGPT across a large, diverse CRC cohort.
Conclusion
To our knowledge, this is the largest study(34,244) to date examining MGPT in CRC, demonstrating high rates of clinically actionable variants detected across all age groups, panel sizes, and racial/ethnic groups. This work supports consideration of broadening germline genetic testing criteria for individuals with CRC.
Treating advanced dMMR Endometrial cancer.
Lynch syndrome is a dominantly inherited cancer syndrome caused by germline pathogenic variants of mismatch repair (MMR) genes. In women with LS, gynaecological cancers are as common as gastrointestinal cancers.
Gynaecological cancer is often the first cancer diagnosis in women with Lynch syndrome.

Reimbursement of new cancer meds is an area where Ireland is increasingly falling behind. Are they expensive? Yes. Is every new ‘innovative’ drug worth its price tag in terms of real world outcomes? No. It’s complex. But now there’s a growing public vs private access divide. @PriscellaLynch
Very informative thread…. https://x.com/mccarthymt7/status/1853202348484747502
“List of anti-cancer drugs that are approved by the HSE for the treatment of endometrial cancer in Ireland. Except for dostarlimab. The protocol was included here on the basis of EMA approval and to offer guidance to private hospitals, where the drug is available for this indication. Probably.”
“Summary: Dostarlimab is very effective in treating advanced dMMR endometrial cancer. The public healthcare system in Ireland cannot afford this drug for public cancer patients (it has been fully assessed by the HSE reimbursement process and declined). Private cancer patients can access this treatment. In public hospitals we instead will continue to use less effective therapies, that are not EMA approved.”
LS-related tumours are characterised by a highly immunogenic tumour-environment that can be targeted by specific immune checkpoint inhibitors.