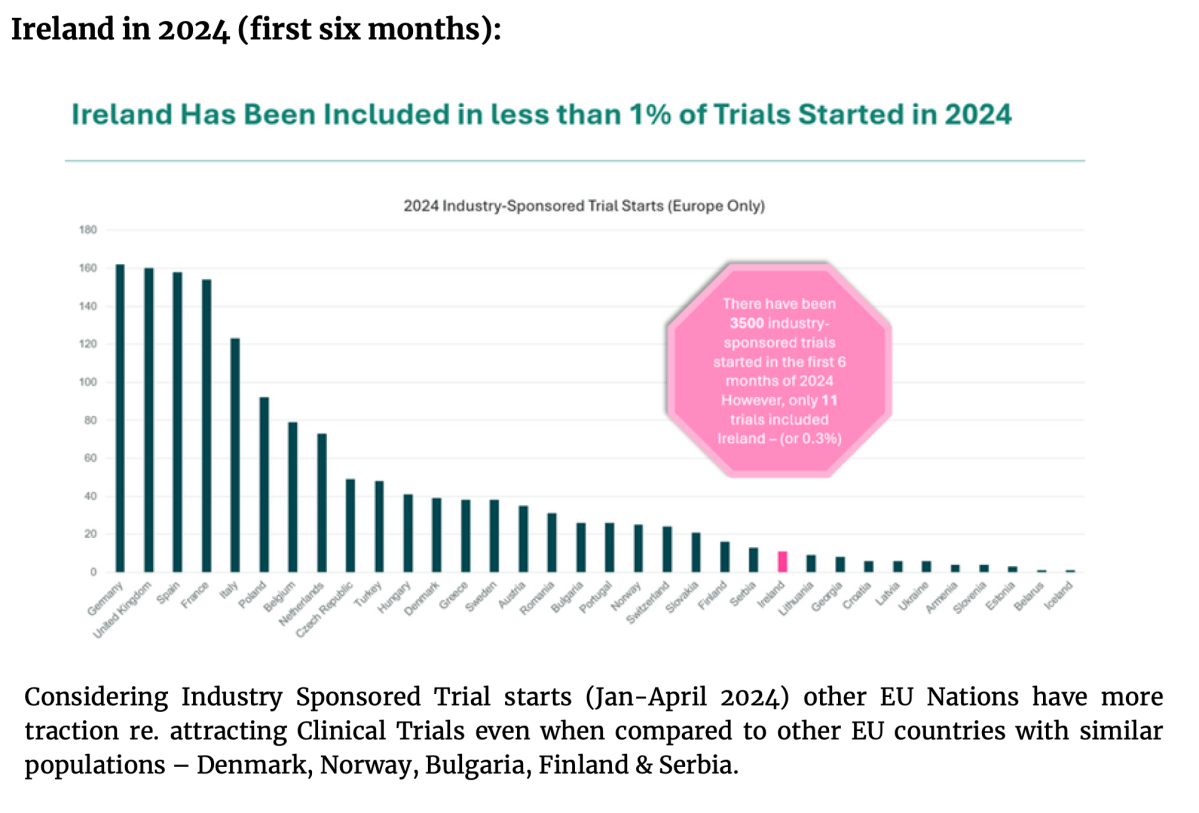
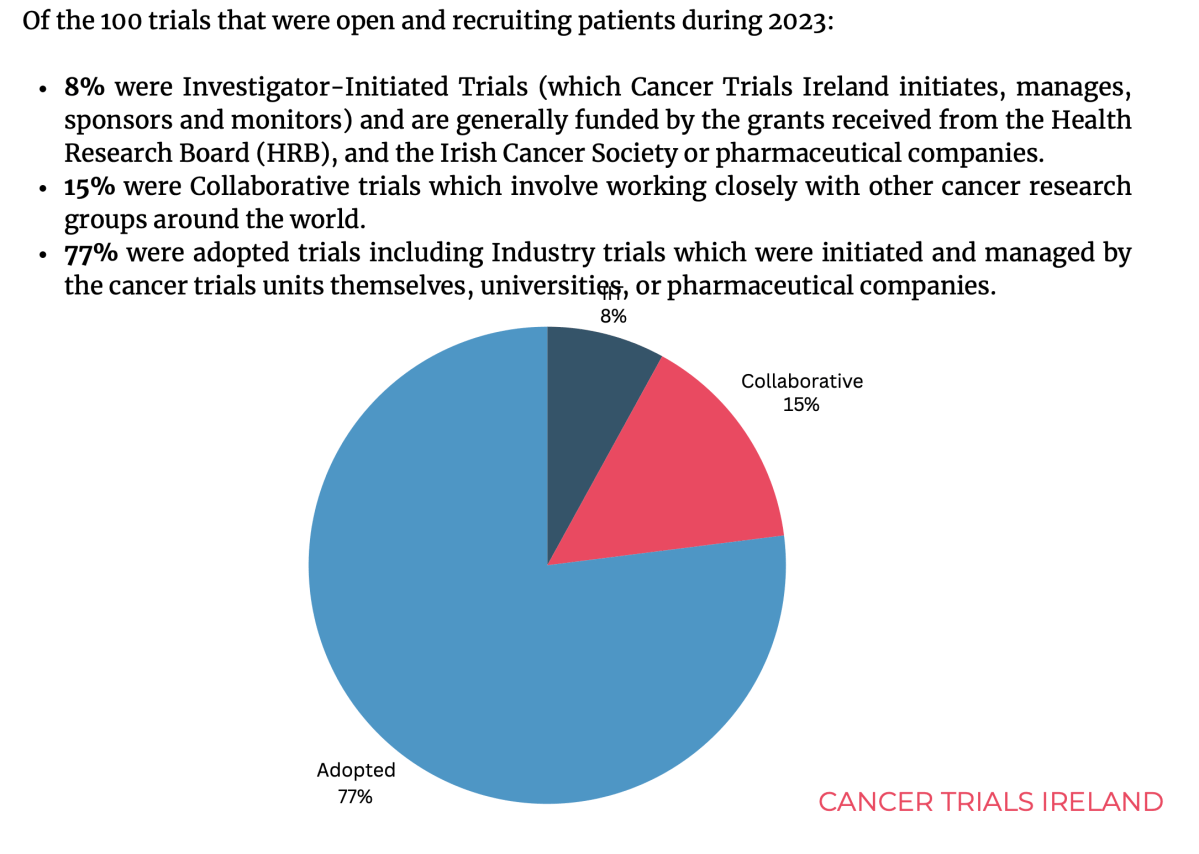
Lynch Syndrome (LS) is a cancer predisposition syndrome caused by constitutional pathogenic variants in the mismatch repair (MMR) genes.
To date, fragmentation of clinical and genomic data has restricted understanding of national LS ascertainment and outcomes, and precluded evaluation of NICE guidance on testing and management. To address this, via collaboration between researchers, the National Disease Registration Service , NHS Genomic Medicine Service Alliances, and NHS Regional Clinical Genetics Services, a comprehensive registry of LS carriers in England has been established.
The most frequently identified pathogenic MMR genes were MSH2 and MLH1 at 37.2% (n = 3362) and 29.1% (n = 2624), respectively. 35.9% (n = 3239) of the ENLSR cohort received their LS diagnosis before their first cancer diagnosis (presumptive predictive germline test). Of these, 6.3% (n = 204) developed colorectal cancer, at a median age of initial diagnosis of 51 (IQR 40–62), compared to 73 years (IQR 64–80) in the general population
The establishment of a secure, centralised infrastructure and mechanism for routine registration of newly identified carriers ensures sustainability of the data resource.
https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(24)00044-0/fulltext