It just might save your life.
During my third colonoscopy colon cancer was discovered and it had advanced to my Lymph Nodes. I had no symptoms.
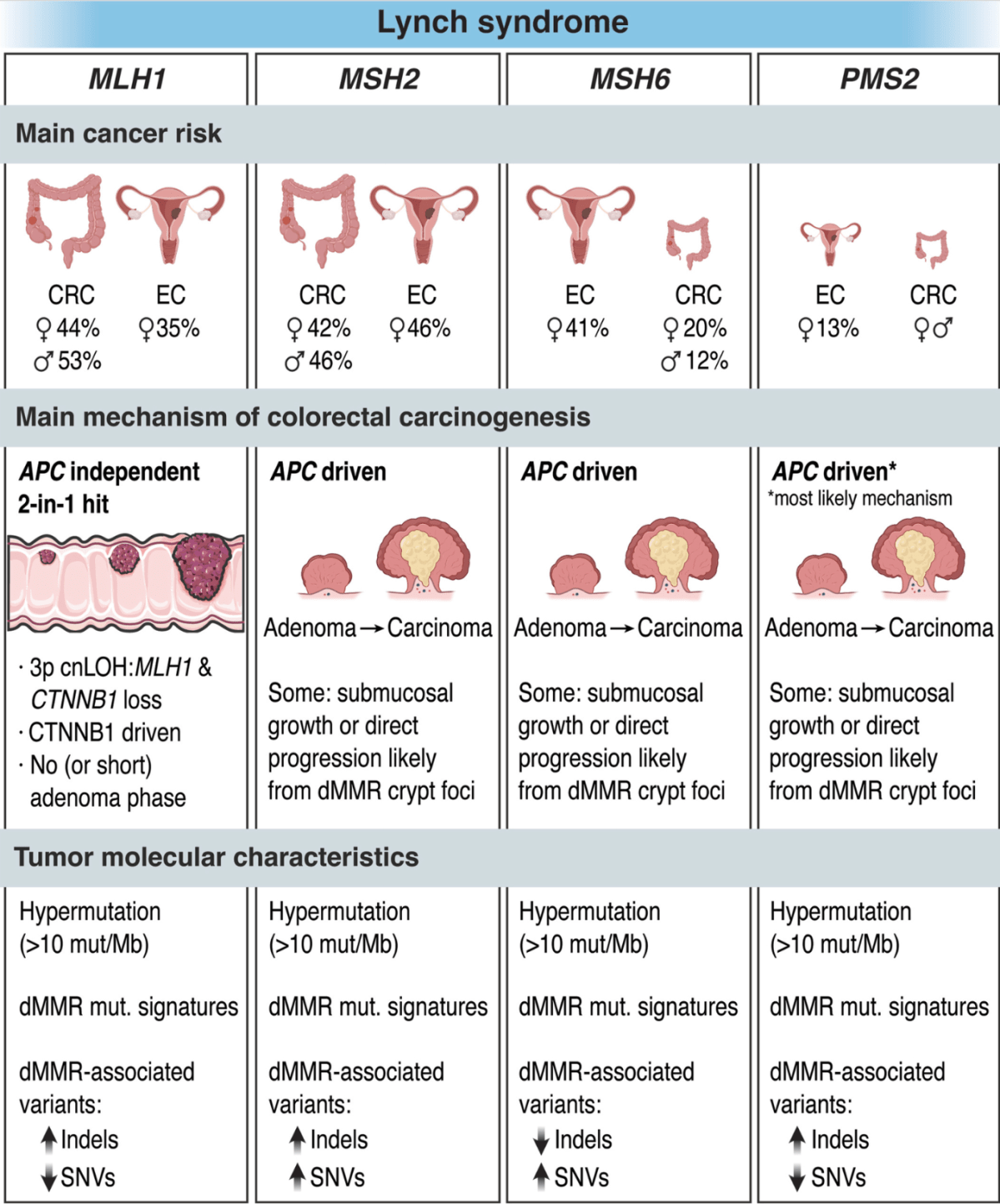
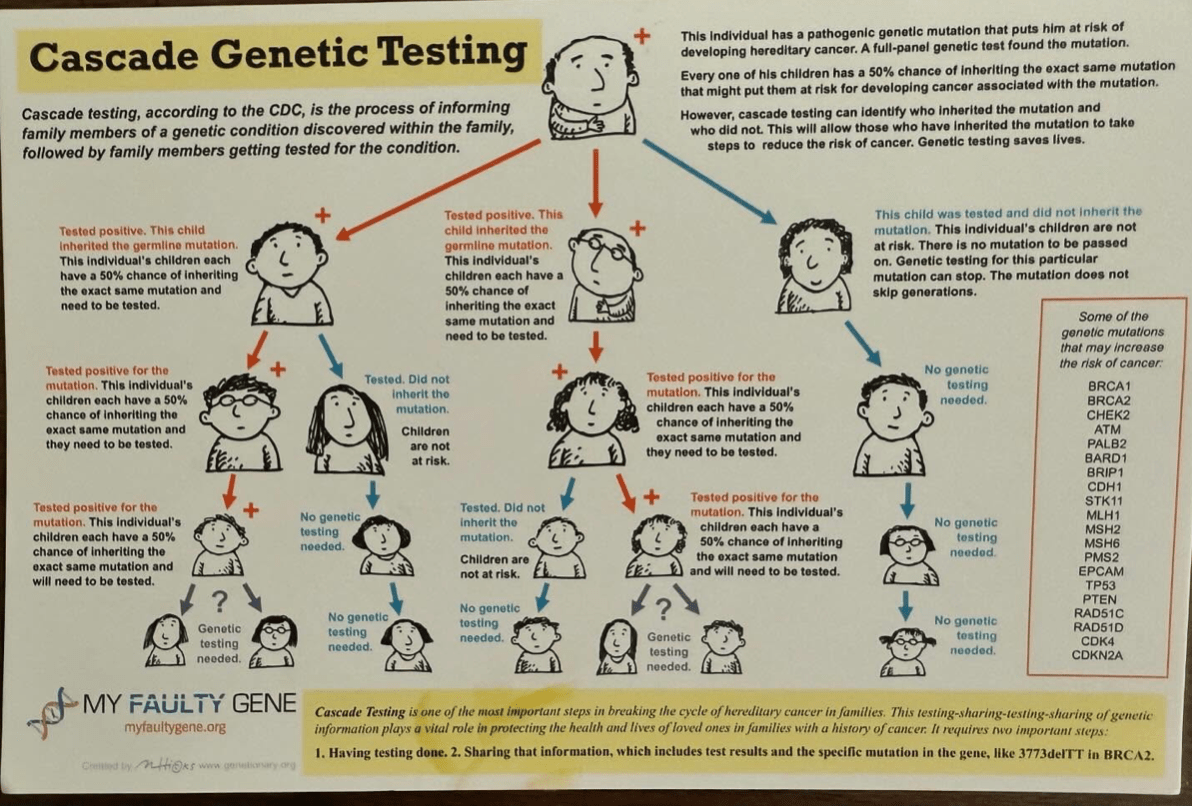
Why was I having colonoscopies? I had a family history of colon cancer and my sibling had tested positive for Lynch syndrome (an inherited predisposition to certain types of cancers including colon and endometrial.
I can understand why many people are nervous about getting colonoscopies. Some wonder how long a colonoscopy will take, how best to prepare for the procedure, how much time you’ll need to take off from work and how embarrassing or disruptive this important cancer screening procedure will be.
Why should people get colon cancer screenings?
The answer is simple: colon cancer screenings can detect colorectal cancer early and prevent unnecessary deaths. Colorectal cancers also are highly preventable and treatable if detected early. In fact, if doctors detect pre-cancerous polyps during a colonoscopy, they can remove the polyps during the procedure.
Prior to Colonoscopy:
Some may think…it’s not pleasant(although having had more than 15 to date I can say I do not find it so, but including the preparation, in most cases you’re spending less than 24 hours to help prevent cancer.
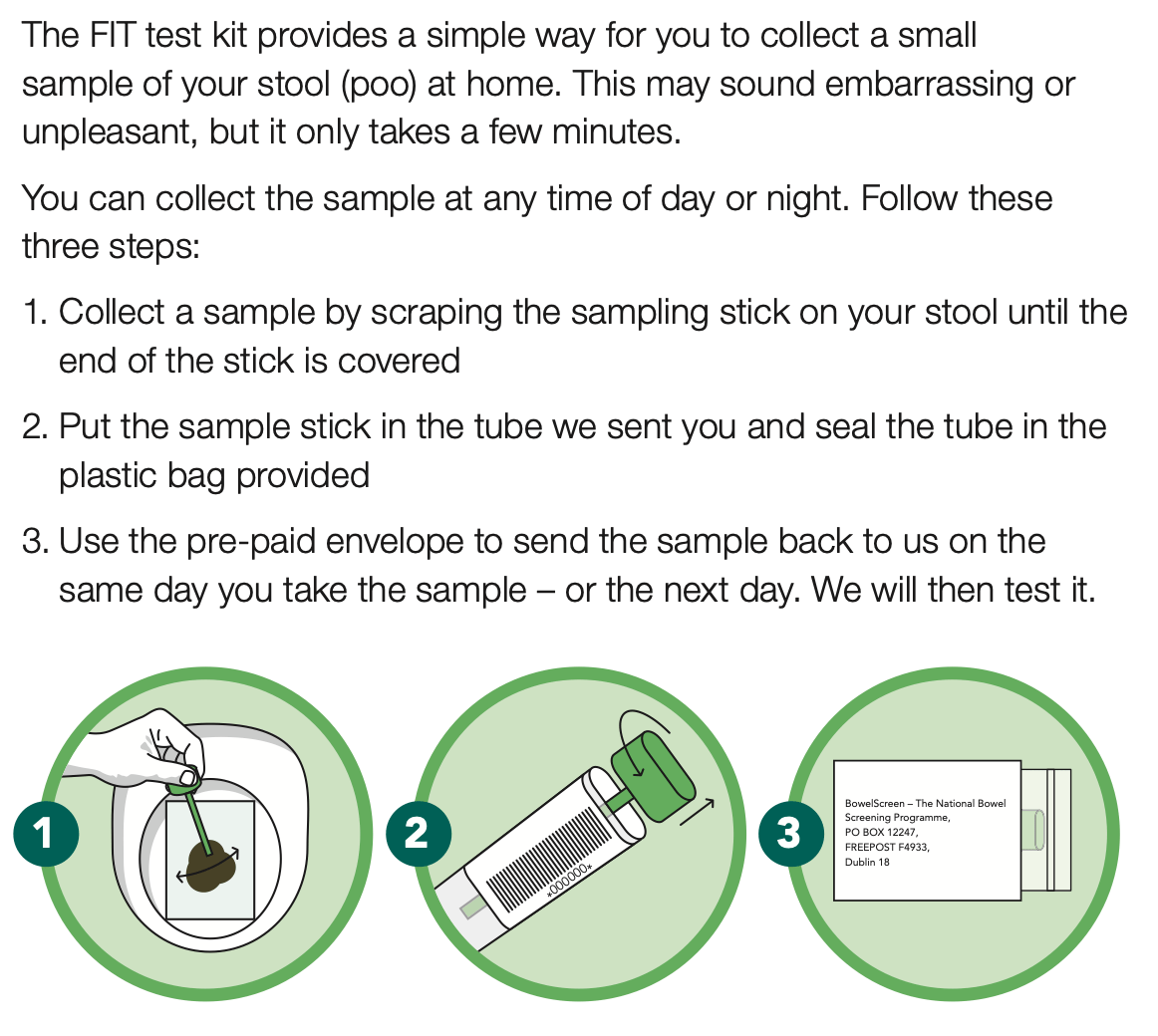
Typically, people prepare for a colonoscopy in their homes the day or the night before along with amending their diet for a few days prior to the procedure. The Bowel preparation prescribed — which involves drinking a lot of water along with prep – can take a number of hours . That’s because you’ll be peeing and pooping out all the waste from your body, so you’ll have a clean, empty colon.
Colonoscopy Procedure:
The actual procedure is quick – usually less than 30 minutes. The doctor inserts a scope into your rectum. You are fully sedated prior to the start of the procedure and you wake up after it is over and you usually don’t remember any of the procedure. For nearly everyone, the actual colonoscopy is painless and relatively quick. You wake up, and you’re done. Aside from not being able to drive themselves home from the procedure, most people feel well and are able to eat and drink normally after a colonoscopy.
What are polyps and how common are they?
Polyps are like skin tags that form on the lining of the colon. Some of them are pre-cancerous, which means that over a period of time, they could become cancerous. So, when polyps are found during a colonoscopy, they can easily remove the vast majority of them. That’s how colon cancer is prevented through colonoscopy.”
https://www2.hse.ie/conditions/bowel-screening/colonoscopy-after-bowel-screening/